Midwifery BACK-TO-BASICS: auditing urinalysis in practice
Sylvia Bone Patient Safety Midwife at West Hertfordshire NHS Trust
The value of urinalysis – setting the scene
Health screening is a crucial part of quality maternal and newborn care (Renfrew et al, 2014). Urinalysis is a useful screening tool that can be utilised throughout the childbearing continuum. It is recommended as a routine investigation in the antenatal period. NICE (2008) recommend testing a woman’s urine at every contact with their midwife. The aim is to screen for a series of infections and/or conditions that could impact on the woman, fetus or new-born’s health and wellbeing.
Conditions screened for through urinalysis include diabetes, pre-eclampsia, dehydration and infections. However, the practice of routine urinalysis has been contentious, especially with regards to screening for pre-eclampsia (Gribble et al, 1995; Hooper 1996; Murray et al, 2002). Yet there remains value for screening alongside blood pressure measurement and also for detecting those women at risk of developing urinary tract infections [UTIs], even if the numbers are small (Murray et al, 2002).
Urinalysis continues to offer a simple dipstick test, with the results produced immediately. If a positive result is identified, at this stage, the urine sample will be sent to the laboratory for further testing that may include a culture and sensitivity or spot protein/creatinine ratio. Additionally, initial urinalysis screening may prompt a series of further investigations and monitoring to support effective diagnosis, treatments and maternity care provision. It is crucial that midwives and maternity care workers have awareness of the value and utility of effective urinalysis to support safe and competent practice.
Urinalysis screening – what are we looking for?
It is important to note that urinalysis is a baseline screening tool. It helps to identify a range of factors, present in the urine that could indicate disease or health issues, each that warrant further investigation or consideration. The following table details the key components of urinalysis that need to be reviewed:
| Component: | Differential diagnosis/cause |
| Blood: | The most obvious reason for blood to be present is the presence of vaginal bleeding however, it can also be a sign of damage in the kidneys, a side effect of anticoagulant treatment and other reasons. (Goddard et al, 2010) |
| Nitrites: | Not usually found but can be present and is associated with Urinary Tract Infection. |
| Leucocytes (White blood cells): | Also associated with UTI. White cells in the urine is known as pyuria or pus in the urine. |
| Protein: | Found in the urine is conditions like hypertension, kidney damage, pre-eclampsia (Mulryan, 2011) |
| Ketones: | This is present where there has been diarrhoea and vomiting leading to fat breakdown. This can also happen in those who are fasting and/or starving and essentially in those with diabetes. In pregnancy, ketones can be found in those with hyperemesis gravidarum. Ketoacidosis occurs due to uncontrolled sugars leading to the blood being more acidic and will need to be reviewed urgently by a doctor (Steggall, 2007) |
| Glucose: | Found in the urine in those with diabetes which is then confirmed by carrying out a GTT as well as those given corticosteroids. |
Childbearing women are at increased risk of recurrent bacteriuria with the incidence of pylenephritis higher than in the general population. Research has identified that those with an abnormal midstream urine sample, in early pregnancy, are more likely to develop a UTI (Murray et al, 2002). Factors associated with a higher risk of bacteriuria in pregnancy include a history of prior UTI, pre-existing diabetes mellitus, increased parity and low socio-economic status (Alvarez et al, 2010; Schnarr and Smaill, 2008; Golan et al, 1989). In part this is due to the pressure the growing fetus and uterus exert on the bladder, in addition to the ureteral dilatation and decreased ureteral peristalsis. The presence of bacteriuria in pregnancy is associated with increased UTIs, preterm birth, intrauterine growth restriction and low birth weight, as such it is important that consideration is given to antibiotic treatment, even for Asymptomatic Bacteriuria (ASB). However, this has led to an increase in the number of broad spectrum antibiotics used to treat suspected and confirmed UTI. The overuse of antibiotics has other public health consequences including pathogen resistance (World Health Organisation [WHO], 2018). To avoid unnecessary treatment, it is crucial that an appropriate urine sample is collected.
Collecting a urine sample for screening – challenges and concerns
The official letter sent out to women inviting them for any appointment, will ask them to bring in a urine sample. As some of the women will not have a sterile urine bottle, the sample is brought in in any container that is at hand. Needless to say, most samples are contaminated, or have sat in their handbags since being collected, usually first thing in the morning which would allow any bacteria to breed and multiply. This would then manifest as having the micro-organisms usually found in a UTI and hence would need sending off to the laboratory for microscopy. Women should provide a fresh sample on site rather than bringing the sample in as this ensures that the sample is collected correctly thus minimising the risk of contamination.
Colour, odour and clarity are all essential indicators in a urine sample and as such it should be a freshly provided sample. In pregnancy, leukorrhea (thin, vaginal discharge) is normal and prevents ascending infection to the uterus (www.nhs.uk, 2018) however, its presence can give false positives. The correct method of collecting a urine sample is to ensure that a midstream specimen of urine is collected avoiding any contaminants. To provide an optimum sample, the woman is advised to first clean her genital area, the first void goes into the toilet, voiding is stopped and a sample is passed into a sterile sample bottle and the remaining void goes into the toilet. This method of start-stop-start is not accurate as sometime too much is voided into the toilet leaving very little to go into the bottle not to mention the difficulty in aiming accurately into the urine bottle with a large bump in the way. The hands usually end up being contaminated with urine which is not a nice situation.
Reflections on practice – conducting a clinical audit
Audit rationale
On joining my current Trust, it became obvious that a high number of urine samples were being sent to the laboratory, on a daily basis, which highlighted the need for a series of audits to explore the clinical impact of this practice. Clinical audits can enhance quality and inform improvements throughout maternity care provision (Byrom, 2017). Two audits were conducted, over two years and the findings are shared here to help maternity units to consider how they might enhance their own service.
Audit aims and objectives
The first audit aimed to review how many urine samples were being sent to the laboratory for culture and sensitivity, the outcomes of these investigations and the costs that this incurred for the women at one maternity service antenatal clinic and maternity day assessment unit.
The second audit aimed to review a new system of urine collection and how this impacted on the urinalysis screening false-positives for contamination and to assess the costs of introducing a new urine collection system across two maternity service clinics.
Audit one methods and findings
Methods
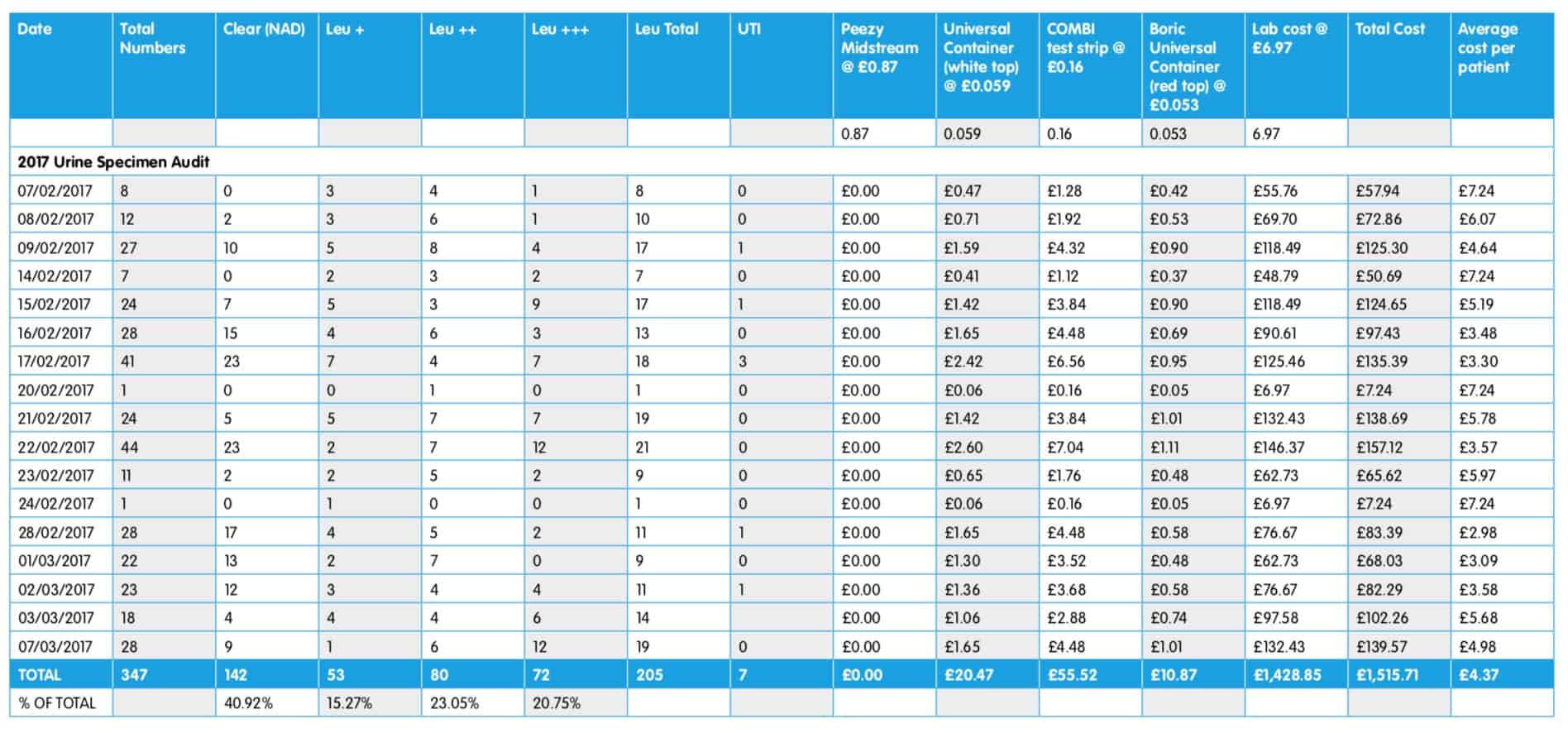
This was a prospective audit of usual practice (Table 2) at one maternity service antenatal and maternity day assessment unit clinic. The audit was carried out Monday to Friday for four weeks during the month of February 2017 (17 working days in total). During this time 427 urine samples were screened using Combi Screen dipsticks and a Urilyzer 100 Pro urometer machine with any positive results being sent on for further laboratory testing.
Table 2 – usual urine testing procedure
| 1. Letter sent to invite childbearing woman to clinic – advised to bring a urine sample with them from home.
2. Urine tested with dipstick strip and urometer at the clinic 3. Any positive results decanted in to red-topped urine sample container and sent to laboratory |
Findings
Of the 347 urine samples screened by urinalysis, 205 were sent off to the laboratory for further testing following positive dipsticks for leucocytes, protein or blood. Out of these 205 samples only 3 were identified as developing e-Coli on culture suggesting a confirmed UTI. A further sample was taken from a women with a known kidney condition. A further 3 samples identified Group B Streptococcus (GBS) infection. The total cost was £1,515.71, with an average patient cost of £4.37.
Table 3– audit one results – clinical findings and costs
The findings of this initial audit were presented at the local clinical and divisional governance meeting and were used to inform staff that urinalysis, following the usual urine testing procedure (Table 2), triggered significant numbers of false-positives and many unnecessary laboratory tests. It prompted the need to review this process and identify a more efficient method of collecting and assessing urine samples. It was decided to repeat the audit once we had identified a new procedure for clean collection of urine samples to assess the impact on both outcomes and costs. This involved reviewing the urine sample collection units used by the local maternity service. The Trust was using a unit called Rocket but we also identified Peezy Midstream which came in a sealed pack containing a wipe to clean the genital area and a plastic funnel to enhance collection of the midstream sample with the initial and final urine being dispensed down the toilet. This Peezy system stops the woman from having to start, stop then start the urine stream whilst ensuring that her hands and the sample bottle remain free from contamination.
Audit two methods and findings
Methods
For the second audit, the initial audit methods were repeated but using the new Peezy system and women were asked to collect the sample at the time of their appointment rather than before. This audit was carried out over a period of 26 working days in which 645 samples were tested.
Table 4– audit two findings
The audit was repeated in January 2018 for 26 working days using the Peezy Midstream device. This second audit found that by collecting a fresh, midstream urine specimen with a Peezy device, there were less false positive samples, and 71.6% were ‘nothing abnormal detected’ [NAD] thus reducing the number sent to the laboratory. Women were much happier as the anxieties associated with UTI were no longer there, and it reduced staff workload as they were not wasting time filling out unnecessary lab forms. This subsequently saved the Trust a lot of unnecessary expenditure as markedly less samples were sent to the laboratory. Of a total of 655 patients, only 186 samples were sent to the lab as the rest were all NAD. The cost to the Trust over 26 days was £1,980.93, with an average patient cost of £3.02.
Comparing results between years and procedures
Reviewing the findings of both audits it can be surmised that using a clean collection system reduced the false-positive results of urinalysis and also reduces the costs of unnecessary laboratory testing. When taking average patient costs into account, we estimated that the monthly direct cost savings were £673.20, and yearly savings of £8,078.43, or £1.34 per patient. Also the Peezy system offers a more efficient and user-friendly unit to aid appropriate sample collection and was found to be valued by staff and service-users alike.
Recommendations for practice:
The following practice points could help you to reconsider your own service provision and approach to urinalysis and screening:
- Conduct an audit of your own urinalysis screening procedures
- Introduce a clean system approach to collecting urine specimens such as Peezy Midstream
- Recommend that women collect their urine sample at the appointment, rather than before
- Ensure women are informed how to collect the sample correctly
References
Byrom, A. (2017). Quality care matters: advancing practice through clinical audits,The Practising Midwife, 20 (3): 18-22.
Goddard, J. et al (2010). Kidney and urinary tract disease. In: College NR et al (eds) Davidson’s Principles and Practice of Medicine. London: Churchill Livingstone
Gribble, RK., Fee, SC., & Berg, RL. (1995). The value of routine urine dipstick for protein at each prenatal visit. American Journal Obstetrics and Gynecology, 173: 214-217.
Hooper DE. (1996). Detecting gestational diabetes and preeclampsia. Effectiveness of routine screening for glucose and protein. Journal of Reproductive Medicine, 41(12): 885-888.
Mulryan C (2011) Urine testing through the use of dipstick analysis. British Journal of Healthcare Assistants;5 (5): 234-239
Murray, N., Homer, C., Davis, G., Curtis, J., Mangos, G. and Brown, M. (2002) The clinical utility of routine urinalysis in pregnancy: a prospective study, Medical Journal of Australia, 177(9): 477-480
NICE (2008). Antenatal care for uncomplicated pregnancies – clinical guidelines CG62, London: NICE. https:// www.nice.org.uk/guidance/cg62/chapter/ guidance#pre-eclampsia
Renfrew, M., McFadden, A., Bastos, MH., Campbell, J., Channon, AA. & Fen Cheund, N. et al. (2014). Midwifery and quality care: findings from a new evidence-informed framework for maternal and newborn care, The Lancet, 384(9948): 1129-1145
Steggall, MJ. (2007) Urine samples and urinalysis. Nursing Standard; 22 (14): 42-45
WHO. (2018). Antibiotic resistance fact sheet, available online: https://www.who.int/news-room/fact-sheets/detail/antibiotic-resistance